Do you need more information



The VIDAS® HIV panel provides a combination of advanced 4th generation assays for improved detection of acute HIV during the critical “window period”.
Do you need more information
Many of the 35.3 million people infected with HIV worldwide are unaware of their condition, as symptoms may be absent or non-specific. In addition, there is a mean period of 3 weeks between contamination and the appearance of the first anti-HIV serum antibodies1. Early detection of seroconversions is thus one of the main HIV diagnostic challenges.
The VIDAS® automated HIV infection screening tests allow simultaneous detection of p24 antigen and anti-HIV antibodies enabling the time lapse between contamination and diagnosis of the infection to be decreased. Identifying more primary infections can make a key difference in reducing transmission. Providing appropriate therapy as fast as possible is also essential to improve patient outcome.
The VIDAS® HIV panel:
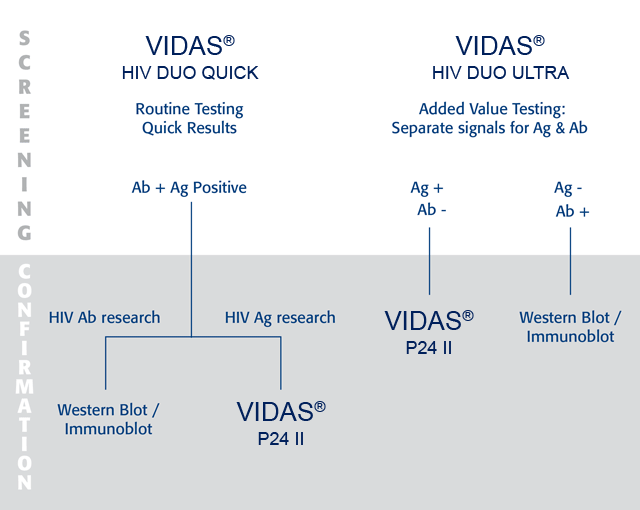
Assay concept:
References:
| VIDAS® HIV DUO Ultra |
VIDAS® HIV DUO Quick |
VIDAS® P24 II | |
|---|---|---|---|
| Reference | 30443 | 30447 | 30117 |
| Tests / kit | 60 | 60 | 30 |
| Sample type | Serum or plasma on lithium heparinate or EDTA | ||
| Sample volume | 200 µL | ||
| Calibration & control stability | 14 days | ||
| Results | Separated results for Ag and Ab |
Global results | |
| Sensitivity in Ag | 11.5 pg/ml HIV Ag | 16.5 pg/ml HIV Ag | 11.25 pg/ml HIV Ag |
| VIDAS® HIV P24 II Confirmation | |
|---|---|
| Reference | 30 444 |
| Tests / kit | 60 tests |
Find more technical details on www.myvidas.com.
Consult your local bioMérieux representative for product availability in your country.
Find more scientific and educational resources on www.myvidas.com.